Sodium Lamp
High lumen output at high efficiency (1920 - Today)

Introduction
HPS Lamps
Inventors of the HPS
Sodium Lamps were first produced commercially by Philips in Holland in 1932. There are two kinds of sodium lights: Low Pressure (LPS) and High Pressure (HPS). These lamps are mostly used for street lighting as well as industrial uses.
The lamp works by creating an electric arc through vaporized sodium metal. Other materials and gases are used to help start the lamp or control its color. See the photos lower on this page for more details.
All credits and sources are located at the bottom of each lighting page
|
LPS
Lamp
|
THE LOW-PRESSURE SODIUM LAMP
The LPS lamp was the first sodium lamp to be developed. It is known by its signature monochromatic yellow color. It is mostly used in Europe since it did not appeal in other markets due to its poor CRI or color rendering. It is among the most efficient lamps in the world because it uses all the current it gets to create light at the most sensitive color (frequency) to the human eye. An incandescent lamp in contrast creates light at all frequencies from Infrared (non-visible) to UV at the other end of the spectrum. The energy used to make non-visible light is a waste of energy since it does not help do the principle job of an electric light. The LPS lamp is also called a SOX lamp (SO for sodium)
Advantages:
- Very efficient lamp
- Powerful lamp for use of large areas
- Despite a warm up time of 5-10 minutes it restarts immediately if there is a brownout
- Lumen output does not drop with age (such as in LEDs or incandescents)
Disadvantages:
- Worst color rendering of any lamp
- Sodium is a hazardous material which can combust when exposed to air (such as if the bulb is broken in the trash)
Statistics:
CRI -44
Color Temperature: 1800
100-190 lumens per watt
Bulb life: 18,000 hrs
Common Uses: Outdoor lighting, security lighting, long tunnel lighting (the light seems to give less fatigue in tunnel driving than white lights flashing by at close proximity).
1. How a modern LPS lamp works
2. Inventors and Developments
1. How the LPS lamp Works
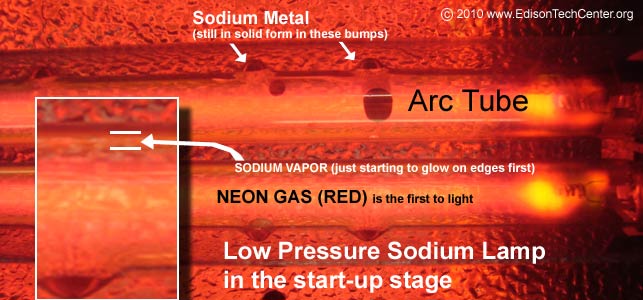
The LPS Lamp is mostly in Europe for outdoor lighting. They create a monochromatic yellow light. In the diagrams below you will see how as it starts it creates a red glow due to the neon gas. The neon gas lights at a lower temperature. As the temperature increases the sodium begins to vaporize and the lamp turns to a pure yellow.


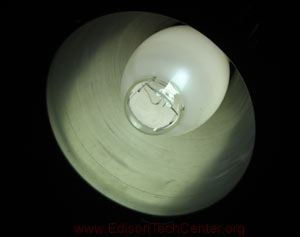
An LPS with it's yellow glow


Argon has a lower glow voltage, argon helps the smaller lamps start at a lower voltage. The larger LPS lamps used in street lighting for the most part do not use argon.
2. LPS Development and Inventors:
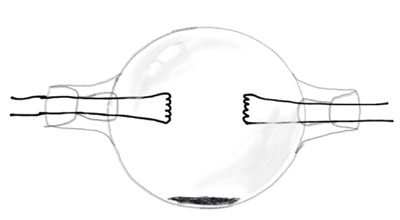
Low pressure sodium lamps were invented first in 1920 by Arthur H. Compton at Westinghouse. The first lamp was a round bulb with two electrodes on each side. The solid sodium metal remained on the bottom center of the bulb. When heated up the metal would vaporize and the lamp would glow yellow. The lamp had to be designed in a sphere because after the metal cooled when the lamp was turned off, the sodium has a property of migrating to the coolest part of the bulb where it solidifies. A tube design would be more particle, similar to the neon lamp which had already been developed by 1920, but it was found that the sodium would migrate to the outer ends of the tube, and there the sodium would destroy the electrodes over time as well as not get hot enough to vaporize. The problem with Compton's models is that the highly corrosive sodium would attack and blacken regular silica glass.
Marcello Pirani led the next major advancement in the low pressure sodium lamp. He worked for Osram in Germany where he developed a sodium-resistant glass in 1931. He used an oven to heat the lamp, which vaporized the sodium and started the lamp.
1932 - The first sodium lamps for commercial sale were made by Philips. Philips has not released the names of the individuals who did the monumental work of developing a reliable sodium lamp ready for widespread use. The first lamps had a removable outer jacket with a vacuum between glass to insulate the bulb to keep it hot enough to keep the sodium in vapor form.
Later developments include:
-Integrating the outer vacuum jacket as a bulb with the inner discharge tube inside, therefore eliminating the separate outer jacket and improving insulation properties.
-Indium tin coating on the inside of the outer jacket to reflect infrared (heat) waves back to the bulb, keeping it warmer and improving reliability in cold weather.
|
|
The HPS lamp is the most ubiquitous lamp for street lighting on the planet. The lamp is an improvement over the LPS lamp in that it has more acceptable color with the great efficiency of the sodium lamp. The better color rendering comes with a bit of sacrifice, it has less efficiency than the LPS. General Electric first developed the lamp in Schenectady, New York and Nela Park, Ohio. The first lamp came on the market in 1964.
 An
HPS lamp with a starting strip, it uses a xenon starting gas. An
HPS lamp with a starting strip, it uses a xenon starting gas. |
|
1. HPS: How it Works
The HPS lamp consists of a narrow arc tube supported by a frame in a bulb. The arc tube has a high pressure inside for higher efficiency. Sodium, mercury and xenon are usually used inside the arc tube. The arc tube is made of aluminum oxide ceramic which is resistant to the corrosive effects of alkalis like sodium.
|
Maintaining a vacuum is difficult, oxygen and other gasses can seep in over time. The getter keeps a stable vacuum by sucking out remaining oxygen and unwanted gasses. The sodium is stored often stored in the amalgam reservoirs on the ends of the arc tube when it is cool unlike the LPS lamp where the sodium is stored in the bumps on the side of the tube (see LPS diagrams)
Disposal:
The Sodium in these lamps is a highly volatile substance. When exposed to air the sodium may explode. The sodium lamp should not be disposed of in normal the normal garbage disposal. There have been many cases of garbage trucks catching fire when the bulbs in the back broke. Sodium lamps also contain mercury. The newer LPS lamps contain less mercury than before, but this has effected performance negatively.
Early inventors of the sodium lamp knew that with a higher pressure in the arc tube that better efficiency could be achieved. The problem was that there was no material that could stand the high pressure, high temperature, and corrosive properties of sodium.
1955
Finally, 35 years after Compton invented the sodium lamp an ideal material was discovered for making an HPS lamp. A researcher named Robert L. Coble working at the General Electric Research Lab near Schenectady, New York developed a material called Lucalox. Lucalox was a commercial term for aluminum oxide ceramic.His work helped pave the way for William Louden, Kurt Schmidt, and Elmer Homonnay to invent the HPS lamp.
1964
Louden, Schmidt and Homonnay worked to create a HPS lamp at Nela Park, Ohio (A General Electric research park in Cleveland, OH). Using the new Lucalox material they figured out how to create an arc tube, evacuate the tube, and insert electrodes that would withstand the hostile conditions inside the tube. The first commercial release of the lamp was in 1964. In the 1980s GE engineers further improved the lamp life and efficiency.

Louden, Schmidt and
Homonnay with a prototype light
Below: video of probably the only remaining 1960s HPS prototypes in the world. It was saved from being disposed of at the GE research lab in Niskayuna, New York.
Lamps are presented in the order of chronological development
Previous: Mercury Vapor Lamps 1901
Next: Fluorescent Lamps 1934
The Electric Light Introduction Page
COMMENTS?
Help us edit and add to this page by becoming a ETC volunteer!
Give us feedback on this and other pages using our Facebook
Page
Written
by M.Whelan with additional research by Rick DeLair
Please contact us if you are a historian and wish to correct or improve
this document.
Sources:
The Subdivision of the Light by Unknown
"A
History of Electric Light and Power" by B. Bowers
Westinghouse lamp catalogues from 1901-1903
Photo/Video
use:
Commercial entities must pay for use of photos/graphics/videos in their
web pages/videos/publications
No one commercial or public is allow to alter Edison Tech Center photos/graphics/videos.
Educational Use: Students and teachers may use photos and videos
for school. Graphics and photos must retain the Edison Tech Center watermark
or captions and remain unmanipulated except for sizing.
Permissions
- Videos: We do not email, FTP, or send videos/graphics to anyone
except in DVD form. Payment is needed for this service. See our donate
page for pricing, and our catalogue
for a listing of videos on DVD.
Professional video production companies may get videos in data form with
signed license agreements and payment at commercial rates.
Copyright
2013 Edison Tech Center